No products in the cart.
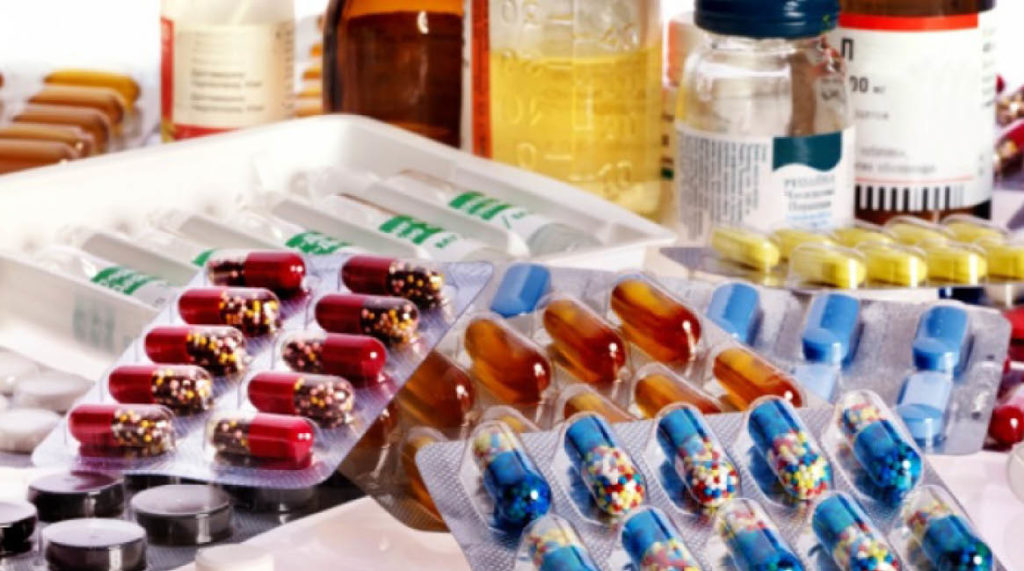
THE Zambia Medicines Regulatory Authority (ZAMRA), says it has intensified surveillance activities across the country, in order to ensure compliance with the recent medicine recall directive and prevent continued circulation of the affected medicines. ZAMRA Public Relations Officer, Ludovic Mwape also notes that it will be conducting follow-up engagements with the suppliers of Deltaprim tablets […]
Subscribe to our newsletter to get our newest articles instantly!
Sign in to your account
Facebook
X.com
LinkedIn
Instagram
Pinterest
More
Social Media
Communication
Bookmarking
Developer
Entertainment
Academic
Finance
Lifestyle
Post Views: 0
You may be interested
“Ruth Grand Phiri and Her Commitment to Transforming Chipata City”
Tuesday, February 10 2026By admin
Ruth Grand Phiri is widely recognized as an inspiring and...
Women and girls in science: Dismantling barriers, closing gender gaps
Thursday, February 12 2026By admin
Facebook Twitter Print Email Although women are more likely than...
UN warns civilians remain at risk as airstrikes continue across Gaza
Thursday, February 12 2026By admin
Facebook Twitter Print Email Fresh airstrikes and shelling across the...